 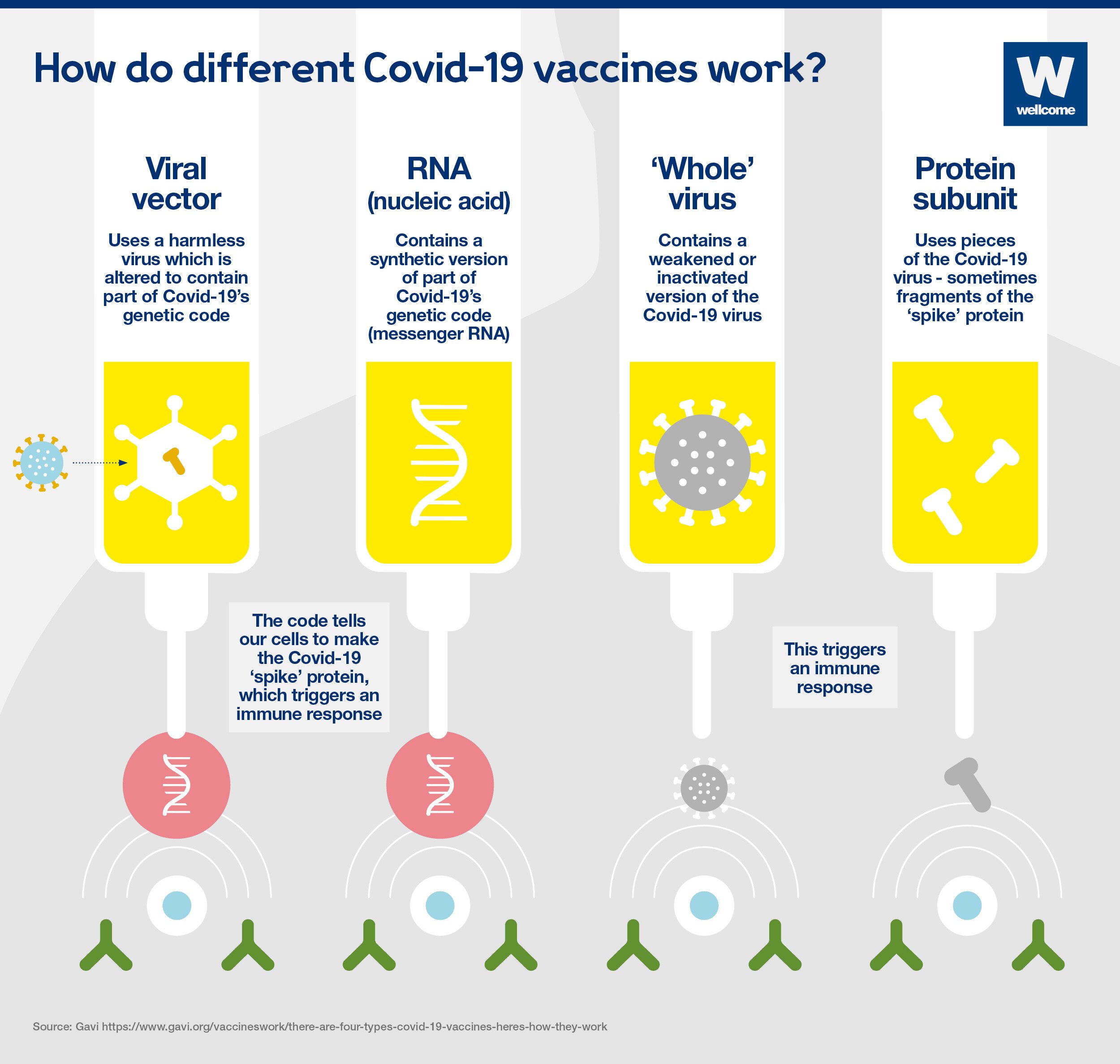 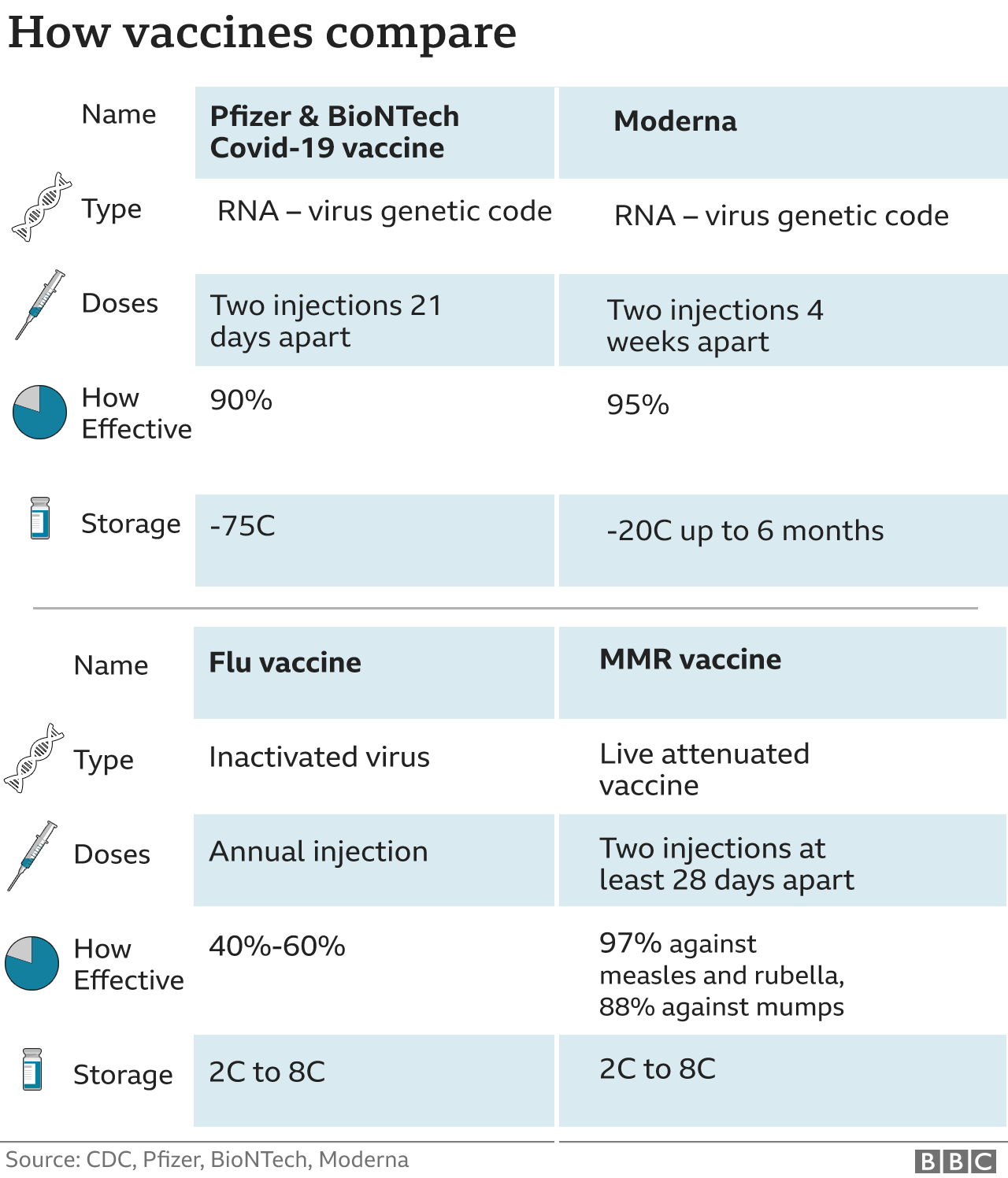
population - around 56 million people - have received Pfizer and Moderna's latest boosters since they were approved in September, according to the CDC. That's slightly lower than the $1.5 billion Novavax's shot generated last year.īut the three companies still face the same hurdle: It's unclear how many Americans will roll up their sleeves to take updated vaccines later this year, even if those shots are delivered on time. The second dose can be given four to eight weeks after the first dose. Call 1-80 Find a vaccine near you You can also check with your local health department or local news for additional information where vaccines are available in your area. The Moderna COVID-19 vaccine is 94 effective in preventing COVID-19 with symptoms. The company hopes to rake in $1.06 billion to $1.24 billion in sales of its Covid vaccine this year. The FDA has approved the Moderna COVID-19 vaccine, now called Spikevax, to prevent COVID-19 in people age 18 and older. The FDA's strain selection positions Novavax as a viable competitor against those household names. Now, one of Novavax's top priorities is to capture commercial market share after lagging behind Pfizer and Moderna. approval for its Covid vaccine under emergency use just last year due to regulatory and manufacturing delays. The jab generated $18.4 billion in revenue last year.įor Novavax, the commercial market is crucial to its survival through 2023 and beyond. The cash-strapped company won U.S. Moderna expects a minimum of $5 billion in revenue from its Covid vaccine, its only available product. Pfizer expects Covid shot revenue to fall to $13.5 billion this year from $37.8 billion in 2022. after reports of rare but serious blood-clotting side effects.įor Pfizer and Moderna, the commercial market is an opportunity to tap into more distribution channels than they did under government contracts.īut both companies still expect Covid-related sales to decline this year as the world emerges from the pandemic and fewer people rely on vaccines and treatments. The company's shots are no longer available in the U.S. That doesn't include Johnson & Johnson, a once-leading Covid vaccine developer. Manufacturers will sell their updated jabs directly to health-care providers rather than to the government. is expected to shift Covid vaccine distribution to the private sector as soon as the fall, when the federal government's supply of free shots is expected to run out. Melinda Wharton, a senior official at the National Center for Immunization and Respiratory Diseases, said at an FDA advisory committee meeting Thursday. Shots targeting XBB.1.5 seem "the most feasible to get across the finish line early without resulting in delays in availability," Dr. Personal Loans for 670 Credit Score or Lower The mRNA (Pfizer, Moderna) and the viral vector gene (J&J) in the vaccine does not enter the cell nucleus where our DNA is. (SOGC) recommend that people who are pregnant or breastfeeding are o ered a complete mRNA COVID-19 vaccine series (e.g., Pfizer, Moderna) at any time during. These spike proteins are ideal targets for vaccines. Coronaviruses, like the one that causes COVID-19, are named for the crown-like spikes on their surface, called spike proteins. Personal Loans for 580 Credit Score or Lower How mRNA COVID-19 Vaccines Work Understanding the virus that causes COVID-19. The company said it is continuing to work to develop an omicron-specific booster.Best Debt Consolidation Loans for Bad Credit However, these data showing that the currently authorized Moderna COVID-19 booster can boost neutralizing antibody levels 37-fold higher than pre-boost levels are reassuring," said Stéphane Bancel, CEO of Moderna. "The dramatic increase in COVID-19 cases from the Omicron variant is concerning to all. The news of Moderna's findings adds more scientific backing to the idea that booster shots are an effective way to offer continued protection against the coronavirus, especially with the growing omicron variant. Research also showed a 100-microgram booster dose gave an even higher level of protection - an 83-fold increase in neutralizing antibody levels, Moderna said. 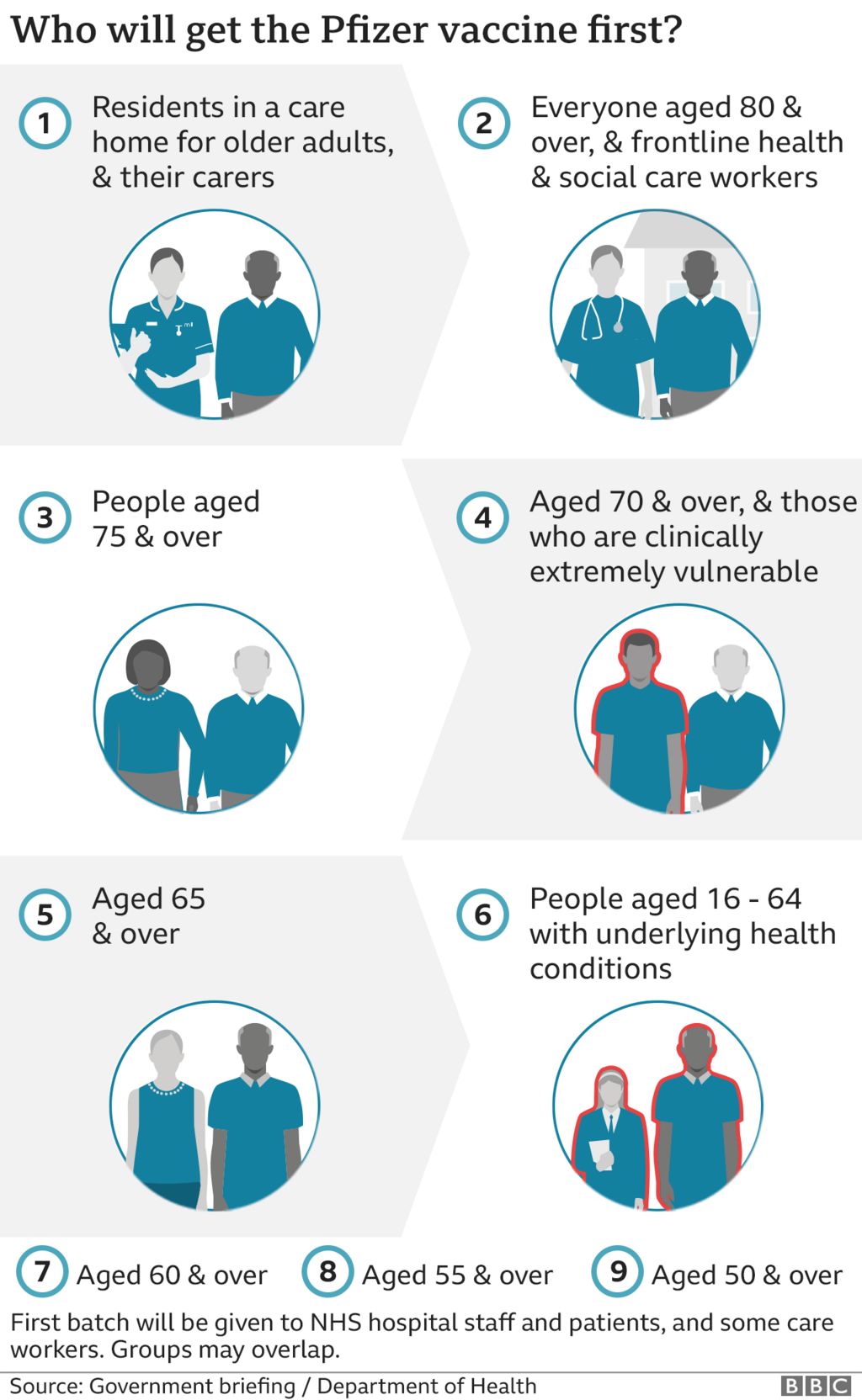
Follow CDC’s and manufacturer’s guidance for vaccine storage. The pharmaceutical company said its current FDA-authorized, 50-microgram booster shot increases antibody levels against omicron 37-fold compared to pre-booster levels. Storage and Handling of Moderna COVID-19 Vaccines. The Centers for Disease Control and Prevention (CDC) continues to emphasize the importance of ongoing surveillance to detect human infections with novel influenza A viruses, including highly pathogenic avian influenza A (H5N1) virus. As downturn fears grow, Bank of America names 8 global stocks to ride out a recession. Moderna said Monday that preliminary data showed that its COVID-19 booster shot is effective against the omicron variant of the coronavirus currently surging in the U.S. 23 hours ago &0183 &32 Here’s where to invest 50,000 right now, according to the pros. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
